Kymera Therapeutics, Inc. KYMR has put up a stupendous efficiency over the previous yr. Shares of this clinical-stage biotechnology firm have surged 82.4% over the stated time-frame in contrast with the trade’s achieve of 17.3%. The inventory has additionally outperformed the sector and the S&P 500 Index throughout this era.
The rally might be attributed to constructive pipeline and regulatory updates for lead pipeline candidate KT-621.
KYMR Outperforms Trade, Sector & S&P 500 Index
Picture Supply: Zacks Funding Analysis
Let’s look at KYMR’s fundamentals, development potential, key challenges, and valuation to make an knowledgeable choice on this context.
KYMR Inventory Surges on KT-621 Examine Information
Kymera is deploying focused protein degradation (TPD) to develop medication for vital well being issues and it has superior the primary degrader into the clinic for immunological ailments.
KT-621 is an investigational, first-in-class, as soon as each day, oral degrader of STAT6, the precise transcription issue liable for IL-4/IL-13 signaling and the central driver of sort 2 irritation.
Final month, KYMR inventory surged after the corporate introduced constructive knowledge from the section Ib BroADen research, which evaluated KT-621 for treating atopic dermatitis (AD), also called eczema.
Information from the research confirmed that remedy with KT-621 led to deep STAT6 degradation in each the 100 mg and 200 mg doses, with median reductions of 94% within the pores and skin and 98% within the blood, exhibiting that the candidate’s results in wholesome volunteers translated nicely to AD sufferers.
Therapy with KT-621 additionally led to robust reductions in disease-relevant sort 2 biomarkers in blood, together with Thymus and Activation-Regulated Chemokine (TARC) – median discount of 74% in sufferers with baseline TARC ranges corresponding to Sanofi’s SNY and Regeneron’s REGN Dupixent (dupilumab) research on AD, Eotaxin-3, IL-31, IgE, and in core sort 2 irritation and AD disease-relevant gene units in pores and skin lesions.
KT-621 demonstrated robust scientific exercise throughout all measured endpoints within the section Ib BroADen research, together with a imply 63% discount in Eczema Space and Severity Index (EASI) and a imply 40% discount in peak pruritus Numerical Ranking Scale (“NRS”).
Therapy with KT-621 was typically secure and well-tolerated, with no critical opposed unwanted effects noticed.
The constructive section Ib BroADen outcomes spotlight KT-621’s potential as a first-in-class, once-daily oral remedy for sort 2 inflammatory ailments. Its week-4 outcomes have been corresponding to—and in some circumstances exceeded—revealed knowledge for SNY/REGN’s blockbuster drug, Dupixent, which is authorised for a number of kinds of inflammatory ailments, together with moderate-to-severe AD.
The FDA additionally granted Quick Observe designation to pipeline candidate KT-621 for the remedy of reasonable to extreme AD.
A section IIb research, BROADEN2, in reasonable to extreme AD sufferers on KT-621 is ongoing, with knowledge anticipated to be launched by mid-2027. Kymera additionally plans to provoke a section IIb research, BREADTH, of KT-621 in bronchial asthma within the first quarter of 2026. These research purpose to hurry KT-621’s growth and decide optimum dosing for upcoming subsequent parallel section III registration research in a number of sort 2 dermatology, gastroenterology and respiratory ailments.
KYMR’s Partnership With Gilead Brings Strategic Advantages
In June 2025, Kymera entered into an unique possibility and license settlement with Gilead Sciences, Inc. GILD to speed up the event and commercialization of a novel molecular glue degrader program concentrating on cyclin-dependent kinase 2 (CDK2) with broad oncology remedy potential in breast most cancers and different strong tumors.
Kymera is advancing preclinical actions for its CDK2 molecular glue program aimed toward treating breast most cancers and strong tumors. If Gilead workouts its possibility, which incorporates an possibility fee to Kymera, it will assume all growth, manufacturing and commercialization tasks for any merchandise ensuing from the collaboration.
KYMR’s Deal With Sanofi
Kymera additionally has a collaboration with Sanofi to advance its pipeline. Nevertheless, in June 2025, Sanofi knowledgeable Kymera that it has chosen KT-485/SAR447971, an oral, extremely potent and selective growth candidate concentrating on IRAK4 for immuno-inflammatory ailments, to advance into scientific research.
The candidate was found by Kymera. KT-485 is being prioritized for growth beneath the businesses’ current IRAK4 collaboration following in depth preclinical work supporting its sturdy growth potential. The candidate is anticipated to advance into early-stage testing subsequent yr.
Consequently, Sanofi discontinued the event of KT-474, which was being evaluated for the remedy of hidradenitis suppurativa and AD in two section IIb dose-ranging research.
KYMR’s Valuation and Estimates
From a valuation perspective, KYMR is overvalued. Going by the value/e book ratio, KYMR’s shares at present commerce at 5.53X, larger than 3.61X for the biotech trade and the corporate’s imply of three.57X.
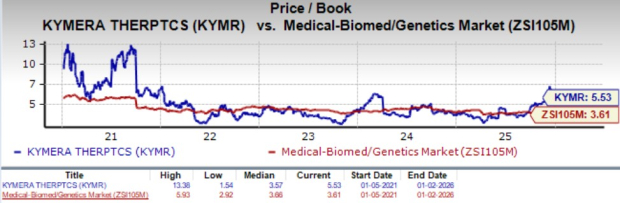
Picture Supply: Zacks Funding Analysis
The Zacks Consensus Estimate for 2025 loss per share has widened over the previous 60 days whereas that for 2026 loss has narrowed to $3.64 in the identical time-frame.
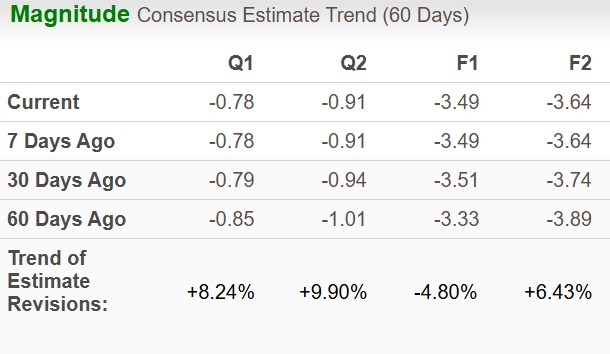
Picture Supply: Zacks Funding Analysis
Keep Invested in KYMR
Kymera’s novel TPD method is promising, and the pipeline progress is encouraging.
Further constructive pipeline updates on KT-621 will likely be a major increase for the inventory. Therefore, we suggest current traders to carry the inventory for now.
Nevertheless, potential traders ought to look forward to a greater entry level. Though the oncology cope with GILD seems promising and the ensuing inflow of money is encouraging, there’s nonetheless a protracted solution to go.
Sanofi’s choice to advance preclinical IRAK4 degrader KT-485 reasonably than advancing KT-474 delayed milestone funds for Kymera that would have been achieved on the approval of KT-474, which had moved to section IIb research in late 2023.
KYMR at present carries a Zacks Rank #3 (Maintain). You may see the whole listing of at this time’s Zacks #1 Rank (Robust Purchase) shares right here.
Zacks’ Analysis Chief Picks Inventory Most More likely to “At Least Double”
Our consultants have revealed their Prime 5 suggestions with money-doubling potential – and Director of Analysis Sheraz Mian believes one is superior to the others. In fact, all our picks aren’t winners however this one might far surpass earlier suggestions like Hims & Hers Well being, which shot up +209%.
See Our Prime Inventory to Double (Plus 4 Runners Up) >>
Regeneron Prescribed drugs, Inc. (REGN) : Free Inventory Evaluation Report
Sanofi (SNY) : Free Inventory Evaluation Report
Gilead Sciences, Inc. (GILD) : Free Inventory Evaluation Report
Kymera Therapeutics, Inc. (KYMR) : Free Inventory Evaluation Report
This text initially revealed on Zacks Funding Analysis (zacks.com).
The views and opinions expressed herein are the views and opinions of the creator and don’t essentially mirror these of Nasdaq, Inc.

